AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 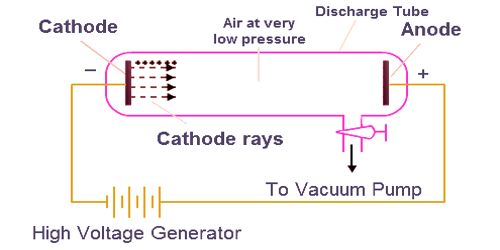
Cathode Ray Tube 12 provides evidence that cathode rays are not visible to human eyes. Do not proceed to schedule a custom demo unless you have already conferred with the lecture demonstrator about it. A series of cathode ray tubes graphically demonstrate various properties of the electron. The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. Thomsons experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. A class activity sheet accompanies this demonstration. The electron beam is deflected by an electric field and a magnetic field. The university shall not be liable for any special, direct, indirect, incidental, or consequential damages of any kind whatsoever (including, without limitation, attorney's fees) in any way due to, resulting from, or arising in connection with the use of or inability to use the web site or the content. Cathode Ray Tube Electrostatic Deflection Tube Physical Electromagnetism Experiment Equipment Middle School Teaching Instrument : Buy Online at Best Price. Cathode Ray Tube 18 - in a darkened room, the electron beam (cathode rays) shows up on a phosphor screen as a bright blue line. However, as shown in the above diagram there was no fluorescent material in the experiment carried out first on the cathode ray tube. cathode-ray tube (CRT), Vacuum tube that produces images when its phosphorescent surface is struck by electron beams. Many websites I read through refer to a fluorescent material. 1.One of the observations I learned was that the glass tube begins to glow with a brilliant green light. The university further disclaims all responsibility for any loss, injury, claim, liability, or damage of any kind resulting from, arising out or or any way related to (a) any errors in or omissions from this web site and the content, including but not limited to technical inaccuracies and typographical errors, or (b) your use of this web site and the information contained in this web site.the university shall not be liable for any loss, injury, claim, liability, or damage of any kind resulting from your use of the web site. Observations in the cathode ray tube experiement. The university expressly disclaims all warranties, including the warranties of merchantability, fitness for a particular purpose and non-infringement. This web site is provided on an "as is" basis. "Do not do demos unless you are an experienced chemist!" Please read the following disclaimer carefullyīy continuing to view the descriptions of the demonstrations you have agreed to the following disclaimer. Clicker questions assess student understanding of how the cathode ray tubes led to scientists understanding of the properties of electrons. A block Power Point presentation used to facilitate the demonstrations and the class activity is available. In addition, the experiment could describe characteristic properties, in essence, its affinity to positive charge, and its charge to mass ratio. Thomson, is one of the most well-known physical experiments that led to electron discovery. The activity sheet has diagrams of each of the cathode ray tubes. CATHODE RAY TUBE, JJ THOMSON EXPERIMENTS AND THE HISTORY OF ELECTRONS - AIS SCIENTISTS. The experiment Cathode Ray Tube (CRT) conducted by J. 
The electron beam is deflected by an electric field and a magnetic field. A class activity sheet accompanies the cathode ray tube demonstrations and experiments. The beams from the guns are manipulated to produce images on a glowing screen. Cathode Ray Tube # 18 - in a darkened room, the electron beam (cathode rays) shows up on a phosphor screen as a bright blue line. Daltons Atomic Theory Thomsons atomic model Thomsons cathode ray experiment The cathode ray tube, also known as the vacuum tube, contains one or more electron guns. However, when cathode rays strike the pinwheel inside the CRT, the pinwheel begins to spin and move. Cathode Ray Tube #16 - light shined on a pinwheel inside a CRT does not cause the pinwheel to move. Cathode Ray Tube #12 provides evidence that cathode rays are not visible to human eyes. Cathode Ray Tube #14 - cathode rays cast a shadow behind a Maltese Cross. A series of cathode ray tubes graphically demonstrate various properties of the electron. The activity sheet has embedded questions designed to promote small group discussions. The diagrams help students understand the significance of each experiment. The activity sheet has diagrams of each of the cathode ray tubes used in the demonstration. 
The typical cold cathode gas discharge X-ray tube found in schools is the. Essentially, our cathode ray tube is just two electrodes in a vacuum chamber with a high voltage applied between them. \): The cathode ray tube was first invented by Sir William Crookes.A class activity sheet accompanies the cathode ray tube demonstrations and experiments. experiments in school science programs involve use of potential sources of. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
